Our view of the Solar System has changed utterly in the last fifty years. Mention that at a cocktail party and your listener will assume you’re talking about Pluto, the demotion of which has stirred more response than any other recent planetary news. But in addition to all we’ve learned through our spacecraft, our view of the Solar System has gone from a small number of orbiting planets to huge numbers of objects at vast distances. Fifty years ago, a Kuiper Belt many times more massive than the main asteroid belt was only theory. And the early Solar System models I grew up with never included any representation of a vast cloud of comets all the way out to 50,000 AU.
We’ve also begun to learn that liquid water, once thought confined to the Earth, may be plentiful throughout the system. Caleb Scharf goes to work on this in a recent post in Life Unbounded, noting what our models are telling us about internal oceans on a variety of objects:
Much can be done with purely theoretical models that seek to determine the appropriate hydrostatic balance between an object’s own gravity and its internal pressure forces – be they from gaseous, liquid, or solid states of matter. Thermal energy from formation, and critically from radiogenic heating (radioactive decay of natural isotopes), all play a role. Throw in a few actual datapoints, measurements of places like Europa or Titan, and these models get much better calibrated. The intriguing thing is that one can play around with compositions and the internal layering of material in a planet-like body to find the best looking fit. As a consequence the nature and extent of any subsurface zones of liquid water can be estimated.
Detecting Interior Oceans
The numbers get to be striking, as Hauke Hussmann and colleagues show in a 2006 paper in Icarus. Start with Galileo, the mission to Jupiter that brought home how much we needed to modify our view of the giant planet’s moons. Galileo discovered secondary induced magnetic fields in the vicinity of Europa, Callisto and Ganymede, offering strong observational evidence for subsurface oceans on all three. The fields are thought to be generated by ions contained in the liquid water layer underneath the icy outer shells. Europa has, of course, become a prime target for future study re astrobiology thanks to the prospect of water combined with a possibly thin ice layer.
The Hussmann paper goes on to calculate interior structure models for medium-sized icy bodies in the outer Solar System, assuming thermal equilibrium between radiogenic heat produced by the core and the loss of heat through the ice shell. Now we really start expanding the picture: The paper shows that subsurface oceans are feasible not just on the now obvious case of Europa, but also on Rhea, Titania, Oberon, Triton and Pluto. A case can also be made for the Trans-Neptunian Objects 2003 UB313 , Sedna and 2004 DW. And note this:
For the bodies discussed here, the liquid layers are in direct contact with the rocky cores. This contrasts with subsurface oceans inside the large icy satellites like Ganymede, Callisto, or Titan, where they are enclosed between ice-I at the top and high-pressure ice layers at the bottom. The silicate-water contact would allow the highly efficient exchange of minerals and salts between the rocks and the ocean in the interiors of those medium-sized satellites.
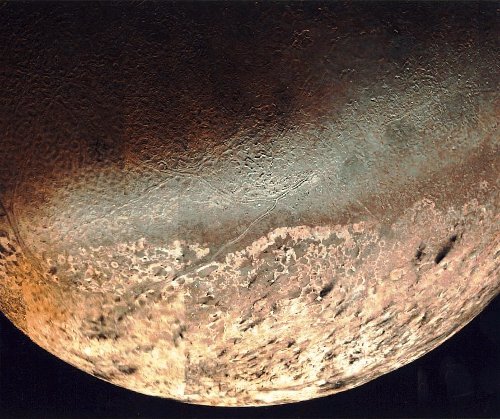
Image: Triton as seen by Voyager 2. Credit: NASA.
Interestingly, given the continued examination of the moon by the Cassini spacecraft, Enceladus does not fit the Hussmann model, the paper noting that sources other than radiogenic heating would be required to sustain such an ocean, the obvious option being tidal heating. We have much to learn about Enceladus (and the paper goes into issues regarding the orbital history of the moon, and the comparison between it and Mimas, where tidal forces are much stronger). But the upshot is clear: We need more observations to confirm whether subsurface oceans are not a common phenomenon in our system’s moons and icy bodies like Trans-Neptunian Objects.
Oceans in the Outer Dark
Hussmann and colleagues assume that subsurface reservoirs on these outer worlds are located beneath a thick ice shell of more than 100 kilometers in thickness — thick enough, in fact, that there is little link between internal oceans and surface features. But study of the interaction between these internal oceans and the surrounding magnetic fields and charged particles, and the response of the objects to tides exerted by the primary, may help us to confirm whether the oceans exist. There’s work here for generations of spacecraft, but get the model right early on and we can make reasonable extrapolations about water’s ubiquity.
The paper’s model, say its authors, is not applicable to Ganymede, Callisto and Titan, but I see that Scharf’s article cites Titan as having possibly ten times the volume of Earth’s oceans in water. This is lively stuff. Quoting Scharf: “…from these bodies alone there could readily be 10 to 16 times more liquid water slurping around off-Earth than on it.” Then factor in those Trans-Neptunian Objects, add the prospect of radiogenic heating, and you wind up with at least the possibility that TNOs could be the largest source of liquid water in the entire Solar System.
Did I say our view of the Solar System has changed? This revolution continues as we push into the Kuiper Belt. New Horizons, it’s hoped, will locate a small Trans-Neptunian Object for study at some point during its journey past Pluto/Charon, but eventually we can hope for the kind of instrumentation around outer planet satellites and other objects that will help us understand their internal composition. If the prospect of internal water bears out on the kind of scales mentioned above, then we have astrobiological potential, even if faint, all the way into the Kuiper Belt.
The paper is Hussmann et al., “Subsurface oceans and deep interiors of medium-sized outer planet satellites and large trans-neptunian objects,” Icarus Vol. 185, Issue 1 (2006), p. 258-273.



Since there are watery oceans sloshing inside those numerous worlds, then there are probably features carved into the mantle derived from ages of hydrological actions. Maybe plate tectonics. Maybe grand canyons. Geysers. Maybe volcanoes and mountains pushed up. Maybe caverns and long passages toward the surface.
I could imagine certain niches for biochemistry to ignite, and persevere in the gloom.
The “water = potential life” meme is being used to excess. Liquid H2O may be necessary, but it is certainly not sufficient. Earth life needs carbon, sulphur and phosphorus, as well as other elements in trace amounts. They also need an appropriate energy source. This is probably going to limit locales for possible life quite drastically. That we are even thinking icy moon oceans as life locales ahead of deep hot biospheres surprises me.
The good news about the ubiquity of H2O throughout the solar system is that the basis for humans greening the system are enhanced. With Titan as a carbon reserve and Io as a sulphur source, if I had to guess, I would say that phosphorus may be the limiting factor, as it is on earth.
Alex, putting aside your premise that phosphorus may be limiting the development of life in other solar system bodies,( wouldn’t all but the lightest element proportions be roughly similar in all larger solar system bodies?) in what way is the available concentration of phosphorus limiting or restraining Earths’ biosphere?
@Mike
Because of the ready availability of nitrogen fertilizers on earth as a result on industrial fixation, Phosphorus becomes the next major limitation. However, Phosphorus generally has to be mined and is in short supply. Even in natural ecosystems, Phosporus is often the limiting factor. In some ecosystems, other elements may be the crucial growth limitations, e.g. iron in open oceans.
The cosmic relative abundance of Phosphorus is far lower than its relative composition in living material, primarily because of Phosphorus’ key role in nucleotides and metabolism, in contrast to the other elements, which led me that that particular assertion. It is just a guess and may well be wrong.
Hi Paul
Since that paper convective transport of dissolved minerals by the high-pressure ice layers has been proposed as one way for the silicates and the oceans to mix. Should be quite efficient, so it’s not a great impediment to very interesting oceanic chemistry.
But oceans of what? Salt water? Dilute sulfuric acid? Magnesium sulfate solution? Ammonia/water mix? Water/methanol mix? We have several often mutually exclusive options.
Water and tides and low gravity should have created many wonders over the long ages at Europa, Titan, Pluto, Triton. I wonder what robot spelunkers might discover out that those worlds. Will there be deep old caverns and long tunnels sheltered from cosmic radiations, perhaps warmed by deep pressures and maybe hot water ? Could any bacteria evolve? Crustaceans? Worms ? Jellyfish?
@Tarmen – Alex Tolley’s comment about life needing an appropriate energy source is spot on. I don’t care if there is are dark oceans of rich tasty soup on every icy rock out to 50.000 AU. Without energetic photons to run photosynthesis, you won’t get far. All the metazoans in hydrothermal vent ecosystems on earth respire – they need dissolved oxygen (from photosynthesis) to utilize those tasty chemosynthetic bacteria. So no crustaceans, no worms, no jellyfish.
PS: The chemosynthetic bacteria also use dissolved oxygen to oxidize hydrogen sulfide. But perhaps a radiogenic process could slowly produce enough free radicals to support some meagre life in a deep dark ocean
If you search on Google “oxygen europa ocean”, you will find a few articles like this, claiming that the oxygen concentration in the ocean could be similar or higher than in Earth’s oceans :
http://www.astronomy.com/en/News-Observing/News/2009/10/Europa%20ocean%20may%20contain%20enough%20oxygen%20to%20support%20life.aspx
See also the Wikipedia article. Europa is a bit of a special case though as it appears to have both a relatively thin crust and be in a high radiation environment to allow for this.
@Joy,
I’d be happy is there were bacteria utilizing ferric ion reduction (or similar) that are found in crustal rocks, so sunlight wouldn’t be a requirement. But clearly multicellular life on earth either photosynthesize or respire aerobically, so some people may be disappointed even if we found life.
While I would be delighted to find life on other worlds in the solar system (the biology would be a scientific bonanza), one concern I have is that like SETI, we will keep looking into ever more unlikely locations. At the same time, we will be demanding spacecraft are sterile and effectively making human exploration almost impossible just in case we contaminate a pristine environment.
What would be really useful is some knowledge of a Gaia hypothesis that ensures that if life exists, there will be an unambiguous binary signature so that planets that look dead really are dead, and vice versa. Perhaps we will get that as we analyze extra-solar worlds.
Oxygen is not an absolute requirement if there are chemical species, out of equilibrium, released from vents etc. Our own muscles generate energy simply from molecular splitting of glycogen.
My main point is this: if you allow that life exists in sub-surface oceans like this, that is quite a game-changer for how we view life in the cosmos. Up until recently, it was all about finding Earth-like worlds in the “habitable zone”. With this new allowance, those considerations go out the window. There is no need for an Earth-like planet and it can be at almost any distance from the star.
Probably 90%+ of stellar systems will have worlds like this. (Our own system has several.) Also, there will be a fair number of free-floater planets of sufficient size to maintain sub surface oceans; there was an article on this topic on here only the other week.
So the new question would have to be, is all life related, because it comes from previous generations of icy moons, or does it originate independently on each icy moon?
Slightly off-topic, but not entirely, since the Habitable Zone concept is closely related to the occurrence of liquid water:
Next Big Future has a very fascinating post about the supposed abundance of habitable planets in our MW galaxy, based on extrapolation of Kepler data:
http://nextbigfuture.com/2011/02/extrapolation-of-kepler-telescopes.html
I agree the most with qraal (= Adam?), that we should not underestimate, but also not overestimate this number, also see ‘our own’ interesting post and discussion on this topic:
https://centauri-dreams.org/?p=11625&cpage=1#comments
@kzb
“if you allow that life exists in sub-surface oceans like this, that is quite a game-changer for how we view life in the cosmos”
But we simply don’t have any information on that. Until we do, it is just speculation on what this might mean. I’d be more inclined to speculate that life exists in Martian crustal rocks, because Mars once had water and Mars and Earth may have exchanged life too, allowing for a single genesis event, rather than two.
If Mars proves sterile, then why should icy moons with oceans be any more attractive?
Remember, we have no definitive evidence of how life started, so we do not know what conditions are required for biogenesis. OTOH, if worlds can swap material and organisms, then it might be possible, even if very unlikely, that Earth could have seeded Europa.
Nice discussion. Just to add a couple points. Recently multi-cellular organisms have been found on Earth that live in entirely anoxic conditions (http://www.biomedcentral.com/1741-7007/8/30/abstract) they simply skirt around the need for dissolved/free oxygen. This doesn’t mean that the broader biosphere could do the same thing, but it’s certainly intriguing. As some of the above comments point out, energy/chemical flux in subsurface environments may be the biggest hurdle for life. Nonetheless, to sustain liquid water (even if full of ammonia or other solutes) requires a thermal input that I think would be comparable to low-energy environments here on Earth. Most microbial life in our planetary subsurface operates on extremely little, with cell-division occurring at rates hundreds to thousands of times slower than in energy rich surface environments. I think the ‘sealed’ environments in our solar system could provide the most stringent test of just how robust the process of ‘origins’ is – does life just pop up in isolation when conditions are right? Or is there more complexity to it?
Alex Trolley:
the key word in “if you allow that life exists in sub-surface oceans like this, that is quite a game-changer for how we view life in the cosmos” is “if”.
I’m not taking any position on the possibility of life on these worlds one way or the other. I’m making the point that if it were true, it would change our whole perception of life. It could have started many billions of years ago in planets and moons right across the galaxy, and been spread around when these got destroyed in supernovae and other mishaps.
One question that seems overlooked in these discussions is: is a world that is perfect for biogenesis, synonymous with the perfect world to nurture life? Has recent work on the potential efficiency of litho-panspermia in transferring endolithic life forms between such worlds meant that we should widen our perspective on this issue?
Okay I admit it, subsurface ocean worlds look just as good cradles of life as Earth to my eyes. Despite the early propaganda, there seems no viable mechanism proposed for the prebiotic Earth whereby Urey-Miller’s soup could be maintained in a high energy state, and it now seems that such a state was either transitory, and rapidly wound down, or the first chemicals for life originated volcanically – either way these would-be paths to life are equally available to the outer worlds as to ours.
Rob Henry, I think we are asking very similar questions. For example, we have now opened up the possibility that life in our system did not start on Mars or Earth, but on some frozen outer world.
There are then the questions, is life in our system the only life, or has it started independently in frozen moons elsewhere? If so is all life related, because it started in previous generations of frozen moons when the universe was young?
Our perspectives might be very similar kzb, but you sneaked in a much earlier origin to life in your comparison, and placed it in the outer worlds. That is very speculative but very interesting. I should criticise it but it brings up so much of interest, such as: the very early appearance of the most advanced type of bacteria alive today in Earth’s fossil record (the cyanobacteria); why life only seems to have appeared once on Earth and its core is all so similar in every single animal; and even possibly why higher forms only appeared after Europa like snowball Earth episodes occurred on Earth (as if the terrestrial inventory of genes worked together better under these conditions).
Those outer oceans are very very old. They have experienced long ages and epochs of vulcanism, chemistry, tides, surprises . They have rhythms of their own. They all have old stories to tell us – the curious new creatures from the Inner Solar System.
Fountains of Optimism for Life Way Out ThereBy GUY GUGLIOTTA
Published: May 9, 2011
For those who hunt for life on other worlds, water in its liquid form is perhaps the leading indicator. Life as we know it on Earth is based on water and carbon. And if organisms can prosper here in nasty environments — in geysers, in the depths of the sea, in toxic waste, in water that is too hot, too cold, too acidic or too alkaline — why could they not prosper out there?
Full article here:
http://www.nytimes.com/2011/05/10/science/space/10water.html?_r=2&pagewanted=1
http://www.technologyreview.com/blog/arxiv/27883/
Soviet Moon Lander Discovered Water on The Moon in 1976
The last Soviet mission to the moon, Luna-24, returned to Earth with water-rich rocks from beneath the lunar surface. But the West ignored the result
kfc 05/30/2012
The possibility of water on the moon has excited scientists and science fiction fans for decades. If we ever decide to maintain a human presence on the moon, clear evidence of water will be an important factor in the decision.
In recent years, that evidence has begun to mount. The data comes from several sources. First there was the pioneering Clementine mission in 1994, America’s first return to the moon in twenty years.
Clementine looked for water by bouncing radio waves off the surface–the returns giving a strong indication that water ice must lie beneath the surface.
Then there was the Lunar Prospector which found a signature for water by measuring the amount of neutrons emitted from the surface and which water ought to absorb).
Then there was Galileo’s flyby of the moon on its way to Jupiter, which also found evidence and more recently, the Indian spacecraft Chandrayaan-I in 2009 which used an infrared camera to spot evidence of water in lunar rocks.
All this has dramatically overturned the previous view that the moon was dry as a bone.
But an interesting question is how this view came to be. After all, there is no shortage of moon rocks on Earth–the Apollo missions brought back some 300 kilograms of the stuff, so much that NASA has lost track of much of it.
Today, Arlin Crotts at Columbia University in New York city throws some fascinating light on this question in a series of three articles about water on the moon and how it got there.
He points out that scientists believed that the Apollo samples were contaminated after their return to Earth.Apparently, the containers used to carry them could not be tightly closed because lunar dust clogged their seals. So any water found in these rocks was thought to have originated here.
What’s more, the Apollo missions confirmed beyond doubt that the river-like channels that earlier spacecraft had seen on the lunar surface were made by flowing lava rather than water. So the prevailing view was that there was that the moon was dry.
However, the Soviets had other ideas. Crotts has unearthed evidence that the Soviets found good evidence of water in moon rocks in the 1970s.
One of the least known missions is the Soviet Luna-24 sample-return mission which landed on the lunar surface in August 1976. This drilled some 2 metres into the lunar surface, extracted 300 grammes of rock and then returned to Earth. An impressive feat by any standards but one that has been largely forgotten in the west.
A Soviet team analysed the sample and found unambiguous signs of water in the rock–they reported that water made up 0.1 per cent of the sample’s mass. In 1978, they published the result in the Russian journal Geokhimiia. This journal also has an in English language version but it was not widely read in the West.
Crott says that today the work has been almost entirely forgotten. “No other author has ever cited the Luna 24 work,” he says.
Curiously, various scientists including the Nobel prize winning chemist Harold Urey, had predicted since the 1950s that water ice and other volatiles ought to be found in craters at the lunar poles, which are permanently in shadow.
Crott goes on to detail a number of other fascinating efforts to find water on the moon, including the famous impact experiment in which NASA slammed an empty rocket stage into one of these shadowy craters to see what the ejecta plume would look like. Sure enough, it contained plenty of water but lots of other stuff too including almost as much carbon monoxide as water.
Today, the idea of a dry moon has been completely overturned. “As recently as 2006 the settled value for the lunar bulk water content was below 1 part per billion. Most values now discussed well exceed 1 part per million,” says Crotts
That’s a remarkable turnaround but one that might have come a little sooner had the Soviet result been taken a little more seriously.
Ref:
Water on The Moon, I. Historical Overview
Water on The Moon, II. Origins & Resources
Water on The Moon, III. Volatiles & Activity