One of these days we’re going to have a new generation of telescopes, some in space and some on the Earth, that can analyze the atmosphere of a terrestrial world around another star. It’s not enough to find individual gases like oxygen and ozone, carbon dioxide or methane. Any of these can occur naturally without ramifications for life. But finding all of these gases in the same atmosphere is telling, because without life to replenish them, some would disappear. Getting the data is going to be hard, which is why new work using the European Southern Observatory’s Very Large Telescope is so interesting.
The work involves ‘Earthshine,’ the reflection of sunlight off the Earth that is in turn reflected off the surface of the Moon. It’s faint, to be sure, but Earthshine is visible in a crescent Moon when the light of the entire lunar disc is visible although only the crescent is brightly lit. Michael Sterzik (ESO) and team have used Earthshine to analyze our own planet’s biosignature, and the results are encouraging. The researchers could deduce from the reflection not only that part of Earth’s surface was covered with ocean, but also that vegetation was present, and both cloud cover and vegetation varied with the rotation of the Earth.
The key is to look not only at brightness variations but at how the light is polarized. This approach, called spectropolarimetry, turns out to be extremely sensitive to biosignatures in reflected light, as co-author Stefano Bagnulo (Armagh Observatory, Northern Ireland) points out:
“The light from a distant exoplanet is overwhelmed by the glare of the host star, so it’s very difficult to analyse — a bit like trying to study a grain of dust beside a powerful light bulb. But the light reflected by a planet is polarised, while the light from the host star is not. So polarimetric techniques help us to pick out the faint reflected light of an exoplanet from the dazzling starlight.”
Polarization tells us more than how bright a given object appears by revealing as well the orientation of the electric and magnetic fields that make it up. Think of the polarized light reflected off a wet road, which polarized sunglasses can reduce by suppressing part of the light (those of us with sensitive eyes rejoice in this fact). The polarized lenses pass only light whose electric vector is in a certain direction. Now we know that the direction of oscillation of the electromagnetic waves we’re studying can be a factor in exoplanet research, not only showing the presence of life but allowing us to separate a planet’s light from that of its host star.
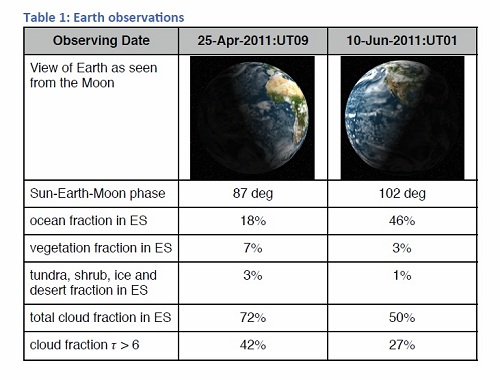
Image: A table from the paper revealing strong biosignatures through spectropolarimetry. Credit: Michael Sterzik/ESO.
The team used the FOcal Reducer/low-dispersion Spectrograph (FORS) mounted at the Very Large Telescope in Chile to measure the linear polarization spectra of Earthshine, comparing its data to models for Earth-like extrasolar planets and also to data from the space-based POLDER (POLarization and Directionality of the Earth’s Reflectances) instrument, for periods in April and June of 2011. While the results are impressive, they may be most significant in helping us tune up our tools. The paper concludes “Improved vector radiative transfer models with more realistic cloud and surface treatment are necessary to fully account for the observed spectra.”
The paper is Sterzik et al., “”Biosignatures as Revealed by Spectropolarimetry of Earthshine,” Nature 483 (01 March 2012), pp. 64-66.



Once again kudos to very, very clever people. The Space Age has been a wonderful time to be alive.
This is the first I’ve ever heard of the possibility of a planet having oxygen without ever having life. Care to elaborate? I will.
In fact, having photosynthesis by itself is not enough to get high oxygen, because the fixed carbon typically sticks around after cell death and soon gets oxidized, with no net addition to the atmosphere in the long run. Only when subduction’s geophysical-scale burial intervenes will O2 climb above even 1%. In our case, the coincident combination of the Gondwana supermountains (20 times the Himalaya range) and the closure of the Iapetus Ocean could provide the stupendous burial rates necessary to get high oxygen. I doubt most terrestrial planets will get so lucky. If it weren’t for that we’d never have gotten past the Ediacarian stage of sponges and other mere metazoans, who unlike true animals can get along fine on 1-5% oxygen.
In fact, it was prior supercontinents that provided the more modest burials that triggered the previous Oxidation Events, all dwarfed by the last one.
So I ask again, how is an always lifeless planet supposed to end up with 20% oxygen?
@Interstellar Bill: I’ve seen it suggested that you can get oxygen atmospheres on runaway greenhouse planets where the water is split into hydrogen and oxygen with the hydrogen being lost to space, or on ocean or supercritical steam planets (especially if the ocean floor is the surface of a high-pressure ice mantle). No idea what kind of oxygen levels are plausible in either case.
Interstellar Bill
Theoreticly a very old planet , staring out comletely covered by deep oceans and with little carbon , could slovly loose hydrogen to space as a result of photodisasociation (and this process could be accelerated by a lack of an earthlike magnetic dield) , leaving oxygen to saturate the general environmet.
Whithout a continous supply of CO2 from volcanoes , no carbonates would settle as limestone , leaving the O2 free to build up. Thats for a non-acidic envoronment ,in a strongly acidic environment it would be much more difficult to predict longrange development .
All the O2 on earth could be produced by an amout of water that would lower the oceans 50 cm.
I am with Interstellar Bill, I cannot figure out how to build a planet with abundant water and little carbon.
This water-dissociation scenario would of course only work on a ‘steam planet’, one of which was just recently discovered. Thus oxygen would still be a sign of life on a non-steam world such as Earth.
@ Ole Burde: interesting, although I would think that CO2 supply to the atmosphere by volcanoes and organic (lime) stone sedimentation, both essential, would be two different and largely independent issues: the first one necessary for replenishment of the atmosphere with new CO2 and hence O2, the second one for C immobilization.
Very impressive achievement.
However: though I have not been able (yet) to look at the paper, I would expect a serious bottleneck with spectroanalysis for biosignatures from exoplanets, even with the planned extremely large telescopes, for one reason: isn’t it so, that for O2, O3, water, …, particularly the infrared part of the spectrum is important? And that part is largely absorbed by our atmosphere.
I have read that this will always be an insurmountable problem with any ground-based platform, even an ELT. And even with adaptive optics.
Is this correct or are there ways around this problem? I would love to be corrected here, i.e. if we could do the future detection of terrestrial planets and their spectroanalysis exclusively with ground-based telescopes. Much cheaper and simpler. Plus: ground-based telescopes with segmented mirrors can in principle be expanded.
Ronald
“interesting, although I would think that CO2 supply to the atmosphere by volcanoes and organic (lime) stone sedimentation, both essential, would be two different and largely independent issues: the first one necessary for replenishment of the atmosphere with new CO2 and hence O2, the second one for C immobilization.”
You got a point there , thats what happen when one tries to explain the world in two sentences… too many loose ends !
Ofcourse its a lot more complicated , there are at least tree major mecanisms for C imobilisation . One is the sedimentatin of limestone , accelerated by shellproducing lifeforms and the other is the acumulation of non-biodigestible C in acidic or oxygendepleted wetlands . The third one is relativly more shortrange , the acumulation og frozen mehtane in permafrost or under great pressure at seabottom . All three forms of imoblisation are ballanced from the release of C by plate tectonics leading to volcanic activity and the gradual release of mostly CO2 .
All this complication somehow leads to a relatively stable and very low concentration of Co2 in a the atmosphere , and it might be this low concentration that would be one of the best sign of life .
Back on our hypothetical oxygen-without-life planet some of the same C imobilisation processes might happen , but thousands of times slower . In order to acumulate an O2 atmosphere without a bigger concentration of Co2 , one solution would be for the the planet to have started out with with little or no C . Another would be a lockdown of platetectonics AFTER al the C had been imobilized . Togeter with the slow pace of fotodisociation it might eventually lead to the folowwing conclution : on an old , waterrich world , an O2 atmosphere will be a much better sign of life , if the Co2 concentration is small but existent .
( still a lot of loose ends ofcourse ! )
Atmospheric chemistry is far too complicated, and the possible variations on it far too little understood, to interpret any simple molecules (such as O2) or even any simple combination of such molecules as “signatures of life.” This kind of hand-waving is readily abused by those who want to find evidence of ET in every poorly understood phenomenon. As an extreme example, why do I keep hearing methane cited as a “signature of life” when it is one of the most common abiotically formed molecules in the universe? It’s a preposterously moronic thing to believe, but alas that doesn’t stop supposedly authoritative “scientists” from declaiming it.
Astronomically more likely, if we observed such simple combinations of simple molecules resembling mixtures in earth’s atmosphere rather than those of other solar system planets, is that they are signatures of chemical processes that don’t much occur with the restricted diversity of planets in our own solar system. We are about to discover many very alien abiotic chemical cycles.
As evidence of life, I want to see spectroscopic signatures of molecules far too complex and thus far too improbable to have assembled without Darwinian evolution. For example, molecules that can accomplish photosynthesis which have, like chlorophyll on earth, covered a substantial fraction of the surface of a planet.
Nick
“……or even any simple combination of such molecules as “signatures of life.”
” signatures of life” is indeed a bad expression , beter to use ” signs of the possibility of life .
As far as I am concerned , we are not talking about any simle combination, but rather trying to buildup a whole profile of parameters for a planet ,including the precense and concentrations of several gases , which seen togeter might be an indication of life .
Forgot the main point again : with ” profile of parameters” the meaning was ofcourse profile of Measurable parameters , such as could be measured with the generation of telescopes coming online in the next few years , more or less the same timeframe as Keplers remaining work . Sadly , none of theese telescopes is designed for such work , but the James Webb telescope might be capable of doing it anyhow .
Above is an interesting claim
“I keep hearing methane cited as a “signature of life” when it is one of the most common abiotically formed molecules in the universe? It’s a preposterously moronic thing to believe, but alas that doesn’t stop supposedly authoritative “scientists” from declaiming it.”
It sounds so reasonable that that premise would lead to that conclusion, that an explanation is in order. Here goes… have you ever tried to make methane by combining hydrogen gas with carbon dioxide, carbonates or carbon monoxide? There’s half the problem, the other bit being that methane is very easily (thus rapidly) broken down in uv light.
Let us not forget that polarized light might give us a handle on specular reflection, which in turn could enable us to map a planet’s surface at high resolution by modeling the path that the “glint-spot” scans across the planetary surface as days and seasons pass. This would mainly be good for distinguishing liquid from solid surface, which is exciting enough, I think.
Eniac
The “glint spot” is one of the important sources of polarized light . Another category of measurements (which includes the glintspot) could be from the exact timeframe where the planet exits or enters the angle of measurable reflextion during a transit event . While passing in or out of transit , in the last few seconds or microseconds there must be a short period , where the light that has passed through the planets atmosphere gets less and less interferrence from the star . To be really efective , this might demand a much more powerfull starshade than the one belonging to the James Webb telescope . A powefull freeflying starshade somewhere like the distance of Mars ,could be a break-through and relatively cheap upgrade to the James Webb telecope , enabling it to play a role it wasnt designed to do…To my taste it was designed basicly to look for “Awe and Wonder” …the great mystery of creation…. cool , but finding or NOT finding life is urgently necesary for a hundred good reasons , while the great mysteries probably wont run anywhere !
“While passing in or out of transit , in the last few seconds or microseconds there must be a short period , where the light that has passed through the planets atmosphere gets less and less interferrence from the star . ”
If I recall correctly this very technique was used recently with the good old Hubble telescope, essentially getting a spectrograph of an exoplanet sunset. They detected large amounts of water in the atmosphere setting off speculation about a big hot waterworld with oceans so deep they have “hot ice” at the bottom. Our blog host was as usual on the job:
https://centauri-dreams.org/?p=21818